Comprehensive training curriculum bridging traditional pharmaceutical regulatory requirements with the rapidly evolving field of Artificial Intelligence and Machine Learning in healthcare.

Uniquely numbered, personal certificate demonstrating GAIP subject knowledge, fully compliant with industry training records and audit requirements.

Access to global competent authorities lists, mapping charts for GCP-to-GAIP equivalents, validation checklists, and downloadable handouts.
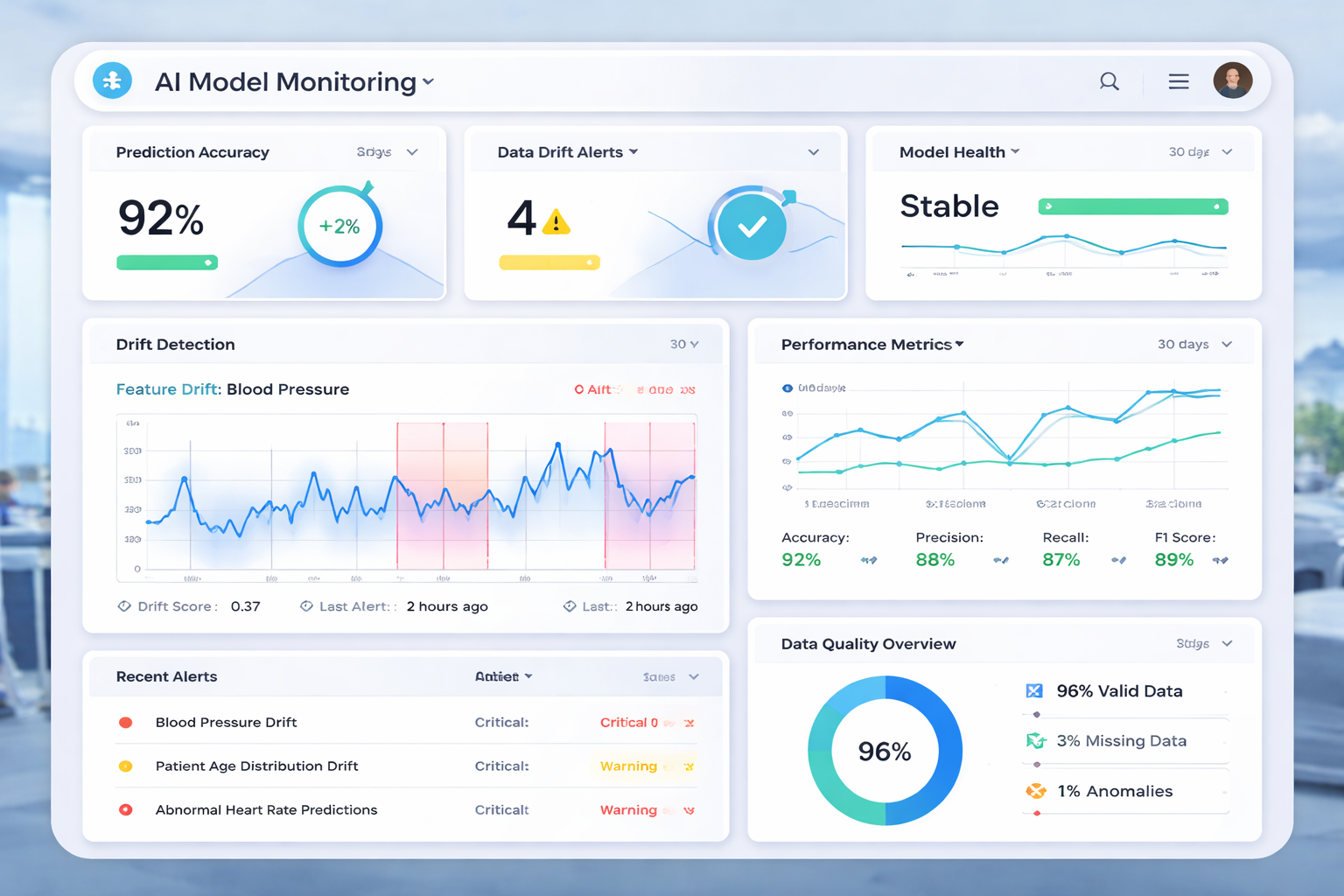
Real-world case studies, monitoring frameworks, drift detection strategies, and hands-on examples from pharmaceutical AI deployments.
Join the next generation of AI-enabled healthcare professionals with comprehensive GAIP certification